This project is now in update mode. Check back regularly to see how things are progressing.
Can Altering Diet Enhance Cancer Therapy
How does the food we eat alter tumor biology? This question is driving us to perform clinical studies to understand and improve our nutrition. Recent data from our (Lew Cantley and Siddhartha Mukherjee) laboratories have:
- demonstrated that diet and metabolism play an active part in determining how cancers grow
- whether they acquire exploitable dependencies and liabilities,
- and whether they become resistant to therapies.
Along these lines, we are conducting clinical trials to assess the ability of a ketogenic diet to enhance the anti-tumor effects of PI3K inhibitors, a class of FDA-approved drugs that slow tumor growth (more details below). While we have already raised over $1.6M to support this work, clinical trials are expensive and we are left to find additional support for staff and state-of-the-art analyses that will be done on the samples we collect.
Why should I help?
Dietary studies are notoriously hard to fund. Despite having an incredibly dedicated team of researchers and impactful pre-clinical data, most sponsors do not support this type of research. There are many reasons why people decide to contribute to cancer research.
- Some donors have personal connections via loved ones and want to make sure others in the future will experience improved outcomes.
- Others just like to promote research that is on the cutting edge and exciting.
Our goal is to create "Precision Nutrition" for cancer, where we formulate diets that explore a deep understanding of the way that dietary nutrients influence cancers. The goal is to match custom-made diets with tumor genotypes and treatment regimens, to improve the outcomes of cancer treatment. Whatever the reason may be, donors are the foundation of our work at Weill Cornell Medicine, and they help us lead the way in innovative treatments and pioneering research.
What is the diet?
Our version of the ketogenic diet is made in the metabolic kitchen at Weill Cornell. It is a type of “classic” ketogenic diet; the one that has been used to treat children with seizure disorders for decades. It is comprised of a 3:1 ratio of (carb+protein): fat, which roughly turns out to be 85% fat, 12% protein, and 3% carb. There is a diverse mixture of fats including those from plants, nuts, and animal products.

The Weill Cornell kitchen is run by a Ph.D.-level dietician who designs and tests the recipes, trains the cooks, and actively manages patients on the diet on a weekly basis. Dr. Hootman's oversight we have been able to keep people with cancer who are participating in a 4-week pilot study, eating 100% of the diet over 4 weeks. This is promising given that published studies using a ketogenic diet in cancer have reported significant dropout rates (30-50%), which limits the ability to detect significant findings.
Why is this a good idea?
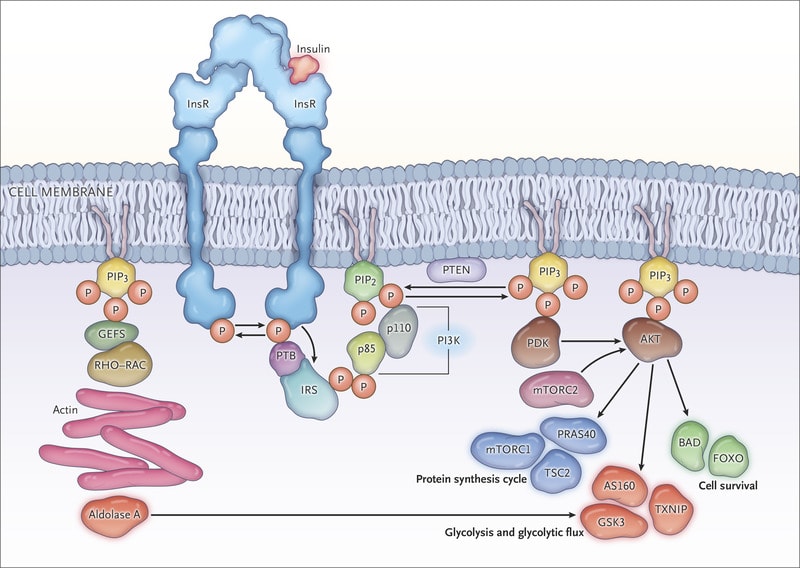
Back in the 1980s, Dr. Cantley discovered an enzyme called PI3K, which is critically important for the response to growth factors in all the cells in our bodies. For example, the hormone insulin is a growth factor that works by stimulating PI3K activity in the muscles and other metabolic tissues. Activation of PI3K in muscles leads to a rise in glucose uptake and enhanced muscle growth. This same process happens in all the body’s cells including cancer cells, if they’re present.
A summary of this process is nicely illustrated in a video that The New England Journal of Medicine (the top medical journal in the world) made for our recent review article. The video can be seen on Instagram.
PI3K is used by every growing cell in the body. Cancer cells can grow uncontrollably, in part, because they developing ways to enhance the activity of PI3K and its partners. Therefore, many pharmaceutical companies have created drugs targeting PI3K (PI3K inhibitors). A few of these drugs are now FDA approved, however there is a problem that limits their use.
The problem?
PI3K inhibitors block PI3K everywhere in the body, not just the tumor. So, after the dose, tissues like muscle can no longer take up glucose and this results in a series of events that causes massive amounts of insulin to be released. Insulin re-activates PI3K and overrides the drug’s effect. We proved this mechanism in a paper that was published earlier this year in Nature, one of the top tier scientific journals.
This work was very well received and has been featured in many news sites (NY Times, Forbes, NY Post, BusinessInsider, Nature article, and Nature commentary )
Keto: the solution
In this study, we showed that a ketogenic diet can protect against the drug-induced insulin release that is described above. This protection makes the PI3K inhibitors work incredibly well in animal models of cancer including pancreatic cancer, which is notoriously hard to treat.
Has this type of study been done before?
No. Despite as many as 48% of people with cancer using popular diets like the ketogenic diet, there has never been a scientific study demonstrating any clinical benefit on people with cancer. In the searchable medical literature, there are about 25 human studies using the ketogenic diet in the setting of cancer. Most of these are very small studies with very variable results. A recent analysis of this data concluded that the available evidence was weak and limited to anecdotal, individual cases. This research has the potential, through diet and nutrition, to further enhance the treatment of cancer.
Listen to the Experts
One Life Podcast: Dr. Lewis Cantley Ph.D. Discusses Sugar and Cancer, Jill Lane and Supplements @28 minute mark
Treating cancer: what role could our diet play? - Science Weekly podcast
Partner Institutions


Lead Photo illustration by Cristiana Couceiro
$50
Level 3
Funds metabolomics (measurement of 275 blood metabolites) analysis for 1 participant for one week
$500
Level 7
Funds the processing, sectioning, and staining of 1 patient's tumor to assess cancer PI3K signaling while on the ketogenic diet
$1,000
Level 8
Funds 1 week of statistics and data analysis to prove the ketogenic diet enhances PI3K therapy
$2,500
Level 9
Funds the measurement of 20 blood cytokines (signals from the immune system) before and after the diet intervention
$10,000
Level 10
creates 3 patient derived cell lines that can be used in future drug screen studies




